Medical Device Single Audit Program
Medical Device Single Audit Program


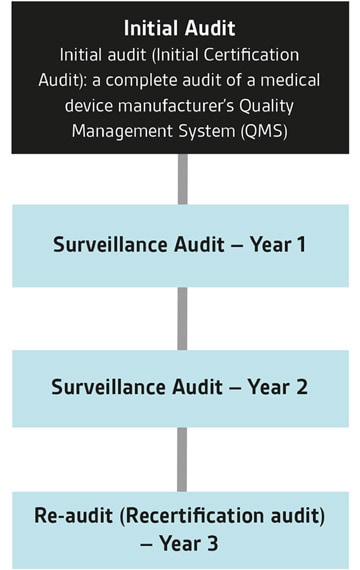
What is a Medical Device Single Audit Program audit?
Through MDSAP, medical device manufacturers need only to be audited once in order to comply with the standard and regulatory requirements of up to five different medical device markets.
These are; Australia, Brazil, Canada, Japan and United States. Audits are conducted by Auditing Organizations (AO), such as BSI, and authorized by MDSAP member Regulatory Authorities (RA). BSI is an Auditing Organization for MDSAP. Here at BSI, we also offer MDSAP in combination with CE, UKCA, ISO 13485 and ISO 9001 assessments. We have been active since MDSAP's inception pilot phase and have already completed a significant number of MDSAP audits, predominantly for world-leading medical device manufacturers and SMEs.